Supporting Evidence Evaluation and Generation in a Swedish Context
An Overview of the Swedish System
Summary
The key actors of the Swedish Health Technology Assessment System include the following: Regional HTA organisations, SBU (Swedish Agency for Health Technology Assessment), TLV (Dental and Pharmaceutical Benefits Agency), and the MTP Council.
Regional HTA centres are linked to healthcare organisations and collaborate through the SBU-chaired HTA Network to streamline methodologies and avoid redundant assessments. SBU performs independent assessments and develops the Method Book for systematic reviews, while TLV evaluates health economics and determines subsidies for medical products. The MTP Council provides recommendations on new medical devices.
HTAs can be initiated by regional HTA organisations through requests from healthcare professionals, MTP Council inquiries, or directly by medical device manufacturers via TLV. The process includes assessments, evidence evaluations, and health economic analyses, culminating in recommendations for decision-making in healthcare organisations.
Key Actors
The figure above details the key actors in the Swedish HTA system. An full screen version of the map can be viewed here. The Kumu map is interactive and allows the selection of individual key actors to access more information and links to additional resources. Here follows a short summary of the system.
Central to HTA, are the regional HTA organisations. These are linked to the healthcare organisations in the regions. Regional HTA centres and regions are represented of the HTA Network, chaired by SBU (The Swedish Agency for Health Technology Assessment and Assessment of Social Services). The aim of the network is to prevent redundant assessments, harmonise methods, and develop new methodologies. SBU carries out independent assessments of methods and interventions in healthcare, among others. The agency also develops the Method Book for systematic reviews and assessments, used by the regional HTA organisations for assessments of medical devices.
TLV (The Dental and Pharmaceutical Benefits Agency) decides on what medicinal products, medical devices and dental care products should be susidised by the state. They also prepare health economic evaluations of evidence on medical devices for decision-making in the healthcare regions.
The MTP (Medical Technology Product) Council has the mandate to issue recommendations on the usage of some new medical devices.
Health Technology Assessment Process Map
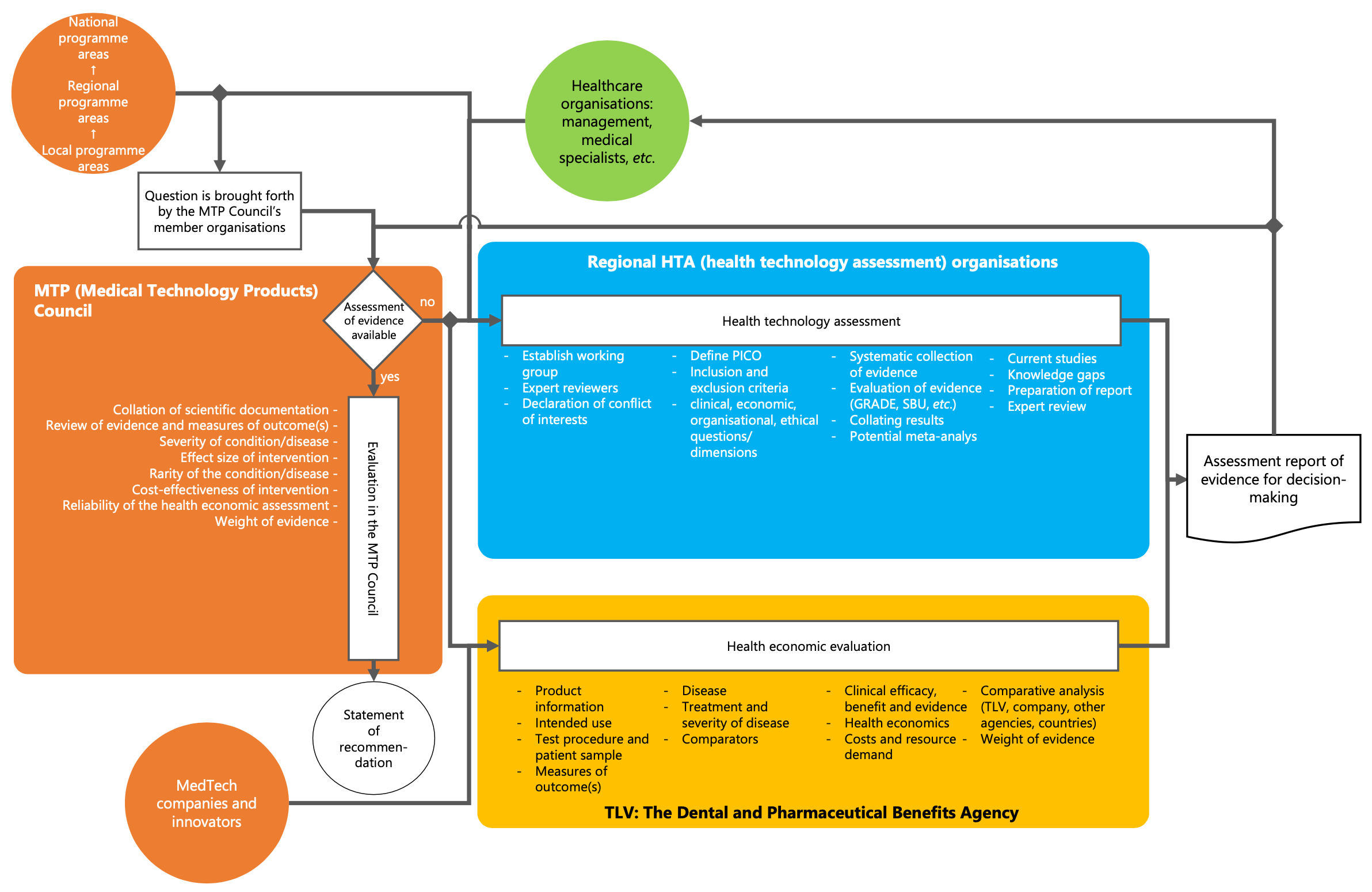
The figure above gives a summary overview of the HTA process; including how the process is initiated, carried out (in general steps) and what the process outcomes are.
HTA can be initiated via several routes, the most common one being through a direct request to a regional HTA organisation from management, specialist consultants or other professionals in the healthcare organisation. Regional HTA is then carried out (see the blue box in the figure below). An assessment report is produced and delivered to the healthcare organisation for decision making. Depending on the state of evidence a mini-HTA may be produced.
HTA can also be initiated through the MTP (Medical Technology Products) Council, where questions are brought by the MTP member organisations (such as, the National, Regional and Local Programme Areas). If an assessment of evidence is available, the evidence will be evaluated to produce a statement of recommendation (see the orange box in the figure below). If no assessment of evidence has been carried out the MTP Council can request one to be carried out, either by a regional HTA organisation or the TLV (the Dental and Pharmaceutical Benefits Agency).
Medical device manufacturers can request a HTA by contacting TLV directly. TLV will produce a health economic evaluation (see the yellow box in the figure below) as basis for decision-making in the regional healthcare organisations or at the MTP council.